Pfizer close to COVID vaccine, Dr. Rodney E. Rohde weighs in on what that means
Staff Contributors | November 10, 2020
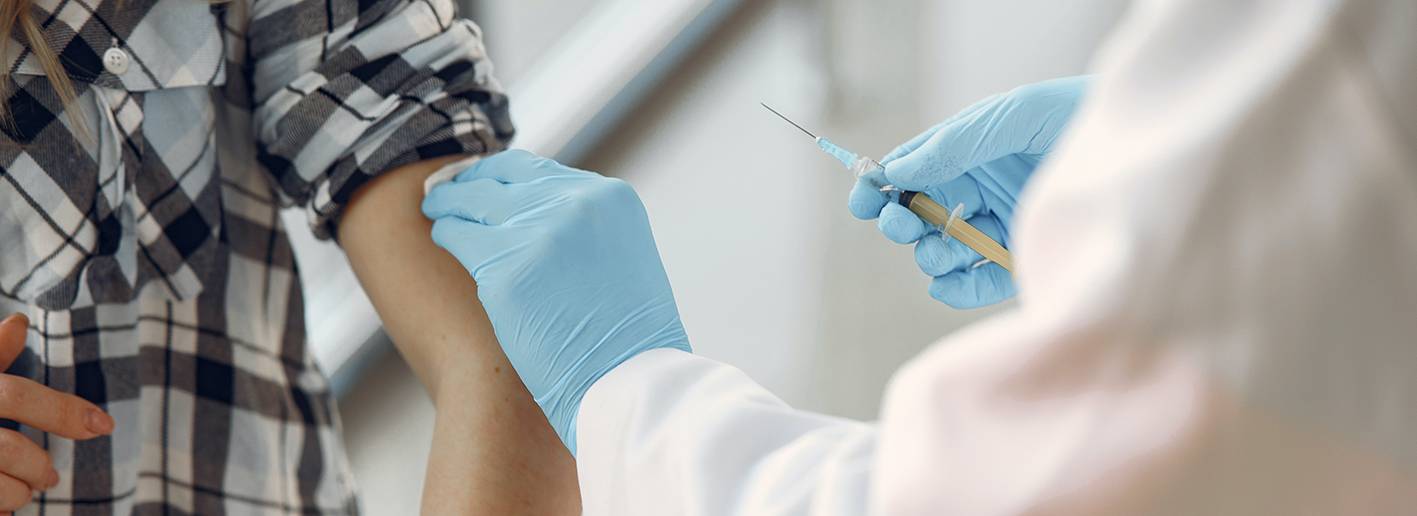

On Monday, Nov. 9, drug maker Pfizer announced that analysis of early data from its coronavirus vaccine may be more than 90% effective. Dr. Rodney E. Rohde, chair and Professor of the Clinical Laboratory Studies at Texas State University, said there is obvious excitement about a possible vaccine in the coming months but urges we still have some critical things to learn.
“While we must always be cautiously optimistic until the Phase 3 trial is complete, this is still great news with a much better efficacy of the mRNA vaccine than expected,” said Rohde, who also serves as the associate director of the Translational Health Research Center. “This type of vaccine technology (mRNA) has never been used before although it was approved. It uses genetic material known as mRNA to basically fool our human cells into creating imposter proteins to mimic what the SARS-CoV-2 virus looks like. In this way, our immune system is tricked into believing the real coronavirus is causing an infection and so it recognizes the imposter proteins as “real” and begins to attack and stop the infection by creating immunological protection,” he said.
U.S. news sources including NBC, CBS, and the New York Post, reported on Tuesday that Health and Human Services Secretary Alex Azar said there will enough vaccine for general public vaccination campaigns by spring 2021.
"This type of vaccine technology (mRNA) has never been used before although it was approved."
“What we do not know yet about the Pfizer vaccine is if it will protect people against all types of the COVID-19 disease from mild to very severe presentation of the infection. Pfizer will also be working to determine if the vaccine will work over years, or if it will only provide seasonal protection (annually) much like the influenza (flu) vaccine. They must also try to understand if the vaccine will protect one against infection even if they have been infected before from the virus,” Rohde said.
Rohde said that the global and U.S. government, public health and healthcare leadership will continue to develop guidelines and plans for utilizing the vaccine in the most effective way possible for the population. “It’s unlikely that there will be enough vaccine to vaccinate every American in a short period of time due to manufacturing processes as well as massive distribution across regions,” he added.
He believes that the U.S. and other countries will distribute the vaccine in a targeted approach starting with the groups who need to be prioritized including and starting with front line healthcare workers, those who have high health risks (immunocompromised) like diabetes, heart disease and other recognized disease risks. The next immediate tier would likely be the elderly (especially those in nursing home and long-term facilities), correctional facilities, and the military or others in roles that place them in high risk categories. The nation’s general population would likely receive the vaccine based on any other tiered approach that is decided to be the best for vaccine stewardship and distribution as well as following the research and healthcare evidence.
According to Pfizer, this vaccine will be free to all American citizens and it was not developed as part of Operation Warp Speed, the US government's coronavirus vaccine effort. However, the U.S. government is investing $1.95 billion to help with making a billion doses or more by next year.
Share this article
For more information, contact University Communications:Jayme Blaschke, 512-245-2555 Sandy Pantlik, 512-245-2922 |